Yttrium Oxide
- Specification
- Data
- Price information
- Video
- Video
- tab 6
- tab 7
- tab 8
- tab 9
- tab 10
- tab 11
- tab 12
- tab 13
- tab 14
- tab 15
- tab 16
- tab 17
- tab 18
- tab 19
- tab 20
Appearance: White solid
Purity: Y2O3/TREO≧99.99%, as per China industrial standard GB/T 3503-2015
Test methods: GB/T 14635 and GB/T 18115.9
Stoichiometric conversion factor: 1.2699
Purity: Y2O3/TREO≧99.99%, as per China industrial standard GB/T 3503-2015
Test methods: GB/T 14635 and GB/T 18115.9
Stoichiometric conversion factor: 1.2699
Chemical formula: Y2O3
CAS No.: 1314-36-9
EINECS EC No.: 215-233-5
HS-Code: 2846901100 (subject to China "dual-use" export licensing)
Industrial standard: GB/T 3503-2015
China export 2024: 2,747 metric tons
CAS No.: 1314-36-9
EINECS EC No.: 215-233-5
HS-Code: 2846901100 (subject to China "dual-use" export licensing)
Industrial standard: GB/T 3503-2015
China export 2024: 2,747 metric tons
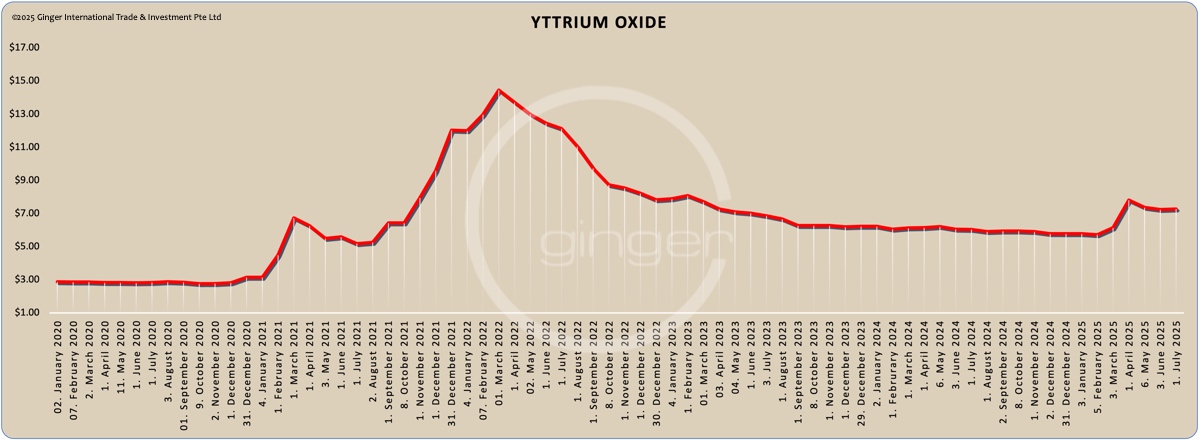
Price level of yttrium oxide on [prices-B1] was ca. [prices-B12]/kg net Ex Works China, incl. 13% VAT.
This is not an offer, it is a general price information. Please use at your own risk.
If you need a specific offer, please contact us and we will be delighted to serve you.
This is not an offer, it is a general price information. Please use at your own risk.
If you need a specific offer, please contact us and we will be delighted to serve you.

