Samarium Oxide
- About
- Specification
- References
- Price information
- Video
- tab 6
- tab 7
- tab 8
- tab 9
- tab 10
- tab 11
- tab 12
- tab 13
- tab 14
- tab 15
- tab 16
- tab 17
- tab 18
- tab 19
- tab 20
Samarium occurs in Samarskite, which is contained in rare earth bearing granite pegmatites.
The main application of samarium is in samarium-cobalt magnets which have permanent magnetization second only to neodymium magnets; however, samarium compounds can withstand significantly higher temperatures, above 700 °C, without losing their magnetic properties. It is also used in lasers as a neutron absorber in nuclear power reactor control rods.
Samarium oxide is also used in infrared and optical glass to absorb infrared radiation. It is a catalyst for alcohols to aldehydes and ketones.
Radioactive isotope samarium-153 is the major component of the drug samarium (153Sm) lexidronam (Quadramet) which kills cancer cells in the treatment of lung cancer, prostate cancer, breast cancer and osteosarcoma. Measurements of samarium and neodymium isotopic ratios are used for geological dating of rocks and meteorites.
The main application of samarium is in samarium-cobalt magnets which have permanent magnetization second only to neodymium magnets; however, samarium compounds can withstand significantly higher temperatures, above 700 °C, without losing their magnetic properties. It is also used in lasers as a neutron absorber in nuclear power reactor control rods.
Samarium oxide is also used in infrared and optical glass to absorb infrared radiation. It is a catalyst for alcohols to aldehydes and ketones.
Radioactive isotope samarium-153 is the major component of the drug samarium (153Sm) lexidronam (Quadramet) which kills cancer cells in the treatment of lung cancer, prostate cancer, breast cancer and osteosarcoma. Measurements of samarium and neodymium isotopic ratios are used for geological dating of rocks and meteorites.
Appearance: yellow-white solid
Putiry: Sm2O3/TREO≧99.5%, as per China industrial standard GB/T 2969-2008
Test methods: GB/T 14635 and GB/T 18115.9
Putiry: Sm2O3/TREO≧99.5%, as per China industrial standard GB/T 2969-2008
Test methods: GB/T 14635 and GB/T 18115.9
Chemical formula: Sm2O3
CAS No.: 12060-58-1
EINECS EC No.: 235-043-6
HS-Code: 2846901940
Industrial standard: GB/T 2969-2008
CAS No.: 12060-58-1
EINECS EC No.: 235-043-6
HS-Code: 2846901940
Industrial standard: GB/T 2969-2008
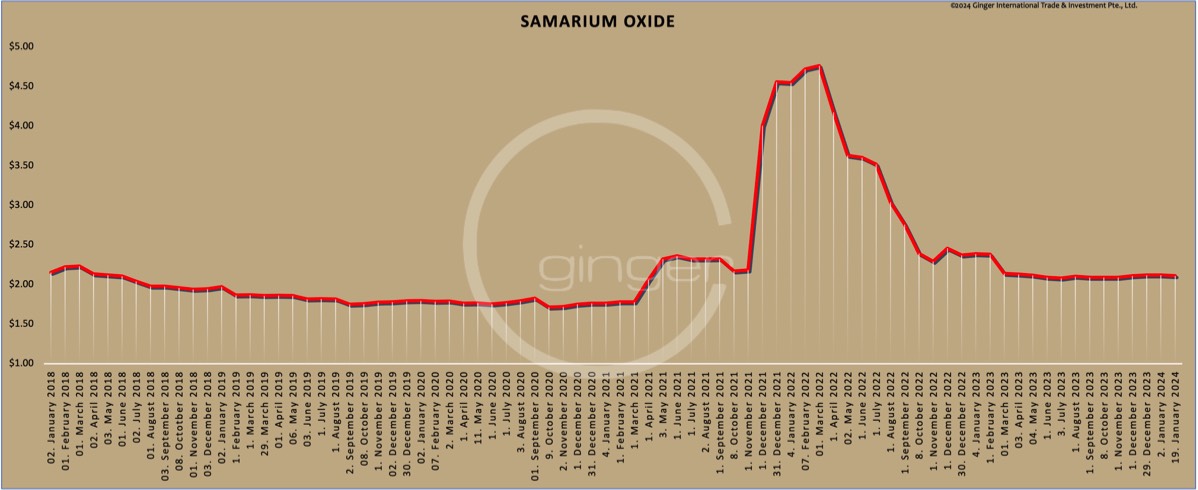
Price level of samarium oxide on [prices-B1] was ca. [prices-B15]/kg net Ex Works China, incl. 13% VAT.
This is not an offer, it is a general price information. Please use at your own risk.
If you need a specific offer, please contact us and we will be delighted to serve you.

